- They are present in all types of cells and all parts of the cell.
- Proteins are the macromolecules responsible for the biological processes in the cell.
- Proteins are polymers of amino acids, compounds consisting of carbon, nitrogen, oxygen, and hydrogen.
- The number of amino acids differs from a few to 3000 or perhaps more in different proteins.
- About 170 types of amino acids have been discovered to take place in cells and tissues.
- Of these, about 25 are constituents of proteins.
- Most of the proteins are nevertheless, made from 20 kinds of amino acids.
Peptide Linkage
Amino acids are linked together to form polypeptides proteins. The amino group of one amino acid may react with the carboxyl group of another releasing a molecule of water. The linkage between the hydroxyl group of the carboxyl group of one amino acid and the hydrogen of the amino group of another amino acid release H2O and C – N link, to form a bond called a peptide bond.
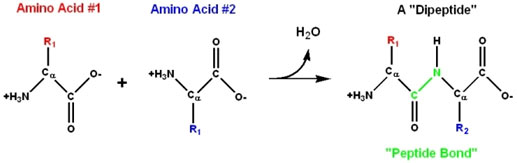
The resultant compound has 2 amino acid subunits and is a dipeptide. A dipeptide has an amino group at one end and a carboxyl group at the other end of the molecule. So, both reactive parts are again offered for additional peptide bonds to produce tripeptides, tetrapeptides, and pentapeptides, etc., causing polypeptide chains.
Structure of Proteins
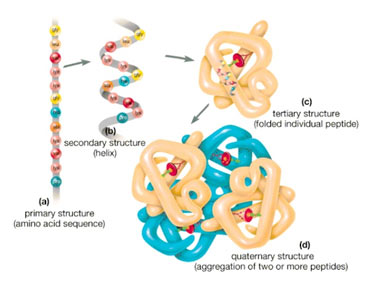
Due to various rearrangement of amino acids, the structure of proteins divides into four types:
- Primary: the covalent linkages of the proteins
- Secondary: the linear peptide chains fold either into an alpha-helical structure(coiled) or a beta-pleated structure(sheets) which contain hydrogen bonds.
- Tertiary: The arrangement and interconnection of proteins into specific loops and bends forms the tertiary structures. This structure consists of hydrogen, ionic, and disulfide bonds.
- Quaternary: this structure is proteins consisting of more than one peptide chain.
Classification of Proteins
Classification of proteins is done on the basis of the following:
On the basis of the Constitution
Simple proteins:
They are composed of just amino acid residue. On hydrolysis, these proteins yield just constituent amino acids.
Conjugated proteins:
They are integrated with non-protein parts. E.g., Nucleoprotein, Phosphoprotein, Lipoprotein, Metalloprotein.
Derived proteins:
They are derivatives or degraded products of simple and conjugated proteins. They may be:
Primary derived protein: Proteans, Metaproteins, Coagulated proteins
Secondary derived protein: Proteosesn or albunose, peptones, peptides.
On the basis of shape
Fibrous proteins:
They include molecules having one or more polypeptide chains in the form of fibrils. The secondary structure is most important in them. They are insoluble in liquid media. They are non-crystalline and are elastic in nature. They perform structural roles in cells and organisms.
Examples are silk fiber (from silkworm, and spiders’ web) myosin (in muscle cells), fibrin (of a blood clot), and keratin (of nails and hair).
Globular proteins:
These are spherical or ellipsoidal due to multiple folding of polypeptide chains. Tertiary structure is crucial in them. They are soluble in liquid media such as salt solution, solution of acids or bases, or aqueous alcohol. They can be crystallized. They disorganize with changes in the physical and physiological environment.
Examples are enzymes, antibodies, hormones, and hemoglobin.
On the basis of nature of Molecules
Acidic proteins: They exist as anion and consist of acidic amino acids. e.g., blood groups.
Basic proteins: They exist as cations and are abundant in basic amino acids e.g., lysine, arginine, etc.
Functions of Proteins
Proteins are vital for the development and growth, and their functions are unlimited. They also have an enormous variety of biological functions and are the most important end products of the pathways.
- Proteins, which are made up of amino acids, serve in many functions in the body (e.g., as enzymes, structural parts, hormones, and antibodies).
- They function as structural elements such as keratin of hair and nail, the collagen of bone, and so on.
- Proteins are the molecular instruments through which genetic details are expressed.
- They execute their activities in the transport of oxygen and CO2 by hemoglobin and unique enzymes in the red blood cells.
- They function in the hemostatic control of the volume of the flowing blood and that of the interstitial fluids through the plasma proteins.
- They are involved in blood clots through thrombin, fibrinogen, and other protein factors.
- They act as the defense against infections by means of protein antibodies.
- They perform hereditary transmission by nucleoproteins of the cell nucleus.
- Ovalbumin, glutelin, and so on are storage proteins.
- Actin, myosin serves as a contractile protein crucial for contraction.
MCQs: Proteins
- What is the primary composition of proteins?
- A) Carbohydrates
- B) Amino acids
- C) Nucleic acids
- D) Lipids
- Answer: B
- What type of bond is formed during the linkage of amino acids to create proteins?
- A) Ionic bond
- B) Covalent bond
- C) Hydrogen bond
- D) Van der Waals bond
- Answer: B
- What is the resultant compound when two amino acids are linked together?
- A) Monopeptide
- B) Dipeptide
- C) Tripeptide
- D) Tetrapeptide
- Answer: B
- In the structure of proteins, what is the role of tertiary structure?
- A) Linear folding of peptide chains
- B) Formation of alpha-helical structure
- C) Specific loops and bends arrangement
- D) Proteins consisting of more than one peptide chain
- Answer: C
- Which proteins are insoluble in liquid media and perform structural roles in cells and organisms?
- A) Globular proteins
- B) Fibrous proteins
- C) Conjugated proteins
- D) Derived proteins
- Answer: B
- What is the category of proteins that include enzymes, antibodies, hormones, and hemoglobin?
- A) Fibrous proteins
- B) Conjugated proteins
- C) Simple proteins
- D) Globular proteins
- Answer: D
- Which proteins exist as anions and consist of acidic amino acids?
- A) Basic proteins
- B) Fibrous proteins
- C) Acidic proteins
- D) Globular proteins
- Answer: C
- What is the primary function of keratin in the body?
- A) Transport of oxygen
- B) Structural element of hair and nails
- C) Defense against infections
- D) Hemostatic control of blood volume
- Answer: B
- In what way do proteins contribute to genetic expression?
- A) Acting as enzymes
- B) Serving as structural elements
- C) Transporting oxygen
- D) Molecular instruments for genetic details
- Answer: D
- Which proteins are involved in the transport of oxygen and CO2 by hemoglobin?
- A) Enzymes
- B) Antibodies
- C) Hemostatic proteins
- D) Contractile proteins
- Answer: C
- What is the main role of thrombin, fibrinogen, and other protein factors in the body?
- A) Blood clotting
- B) Defense against infections
- C) Hemostatic control
- D) Oxygen transport
- Answer: A
- Which proteins act as a defense mechanism against infections?
- A) Storage proteins
- B) Contractile proteins
- C) Antibodies
- D) Nucleoproteins
- Answer: C
- What is the term for proteins consisting of more than one peptide chain?
- A) Simple proteins
- B) Conjugated proteins
- C) Derived proteins
- D) Quaternary proteins
- Answer: D
- How are fibrous proteins characterized?
- A) Spherical or ellipsoidal
- B) Insoluble in liquid media
- C) Soluble in salt solution
- D) Crystalline in nature
- Answer: B
- Which type of proteins are soluble in liquid media and can be crystallized?
- A) Fibrous proteins
- B) Globular proteins
- C) Conjugated proteins
- D) Simple proteins
- Answer: B
- In the classification of proteins based on shape, what is characteristic of globular proteins?
- A) Insolubility in liquid media
- B) Linear peptide chains
- C) Spherical or ellipsoidal shape
- D) Fibrils structure
- Answer: C
- What are the integrated proteins with non-protein parts called?
- A) Simple proteins
- B) Conjugated proteins
- C) Derived proteins
- D) Quaternary proteins
- Answer: B
- Which proteins are derivatives or degraded products of simple and conjugated proteins?
- A) Simple derived proteins
- B) Secondary derived proteins
- C) Primary derived proteins
- D) Quaternary derived proteins
- Answer: B
- What is the category of proteins abundant in basic amino acids?
- A) Acidic proteins
- B) Basic proteins
- C) Fibrous proteins
- D) Globular proteins
- Answer: B
- What are the constituents of proteins made from?
- A) 25 types of amino acids
- B) 50 types of amino acids
- C) 170 types of amino acids
- D) 3000 types of amino acids
- Answer: A
- Which structure of proteins involves hydrogen, ionic, and disulfide bonds?
- A) Primary structure
- B) Secondary structure
- C) Tertiary structure
- D) Quaternary structure
- Answer: C
- How many amino acid subunits does a dipeptide have?
- A) One
- B) Two
- C) Three
- D) Four
- Answer: B
- What is the role of proteins in the hereditary transmission by nucleoproteins of the cell nucleus?
- A) Structural support
- B) Defense mechanism
- C) Molecular instruments for genetic details
- D) Contractile function
- Answer: C
- Which type of proteins perform a crucial role in contraction, such as actin and myosin?
- A) Enzymes
- B) Contractile proteins
- C) Storage proteins
- D) Antibodies
- Answer: B
- What is the function of proteins in the transport of oxygen and CO2 by hemoglobin and enzymes in red blood cells?
- A) Hemostatic control
- B) Blood clotting
- C) Defense against infections
- D) Oxygen and CO2 transport
- Answer: D
- Which proteins are involved in blood clotting?
- A) Hemostatic proteins
- B) Enzymes
- C) Antibodies
- D) Contractile proteins
- Answer: A
- What is the primary function of proteins like ovalbumin and glutelin?
- A) Structural support
- B) Defense against infections
- C) Storage proteins
- D) Contractile function
- Answer: C
- What is the basis of classification for simple proteins?
- A) Constitution
- B) Shape
- C) Nature of molecules
- D) Solubility
- Answer: A
- Which proteins perform as molecular instruments through which genetic details are expressed?
- A) Enzymes
- B) Structural proteins
- C) Nucleoproteins
- D) Storage proteins
- Answer: C
- How much percentage of the total dry weight of cells do proteins comprise?
- A) Less than 10%
- B) Around 25%
- C) Over 50%
- D) More than 75%
- Answer: C
FAQs: Proteins
- What are proteins, and why are they considered the most abundant organic compounds in cells?
- Proteins are macromolecules responsible for biological processes, comprising over 50% of the total dry weight in cells. They are present in all types and parts of the cell.
- What is the composition of proteins, and how many types of amino acids are commonly found in them?
- Proteins are polymers of amino acids, consisting of carbon, nitrogen, oxygen, and hydrogen. While about 170 types of amino acids exist in cells, proteins are mainly made from 20 kinds of amino acids.
- How are amino acids linked together to form polypeptides, and what is the resulting compound of this linkage?
- Amino acids are linked through a peptide bond, where the amino group of one reacts with the carboxyl group of another, releasing water. The resulting compound is a dipeptide, with reactive parts for further bonding to create polypeptide chains.
- What are the four types of protein structures based on the rearrangement of amino acids?
- The four types are:
- Primary structure: Covalent linkages of proteins.
- Secondary structure: Linear peptide chains folding into alpha-helical or beta-pleated structures with hydrogen bonds.
- Tertiary structure: Specific loops and bends arrangement with hydrogen, ionic, and disulfide bonds.
- Quaternary structure: Proteins consisting of more than one peptide chain.
- The four types are:
- How are proteins classified based on their constitution, and what are the main categories?
- Proteins are classified into simple proteins (composed of amino acid residues), conjugated proteins (integrated with non-protein parts), and derived proteins (derivatives or degraded products).
- What distinguishes fibrous proteins from globular proteins based on shape?
- Fibrous proteins have one or more polypeptide chains forming fibrils, are insoluble, non-crystalline, and elastic, performing structural roles. Globular proteins have spherical or ellipsoidal shapes, are soluble, and undergo crystallization, including enzymes and hormones.
- How are proteins categorized based on the nature of molecules, and what are examples of each category?
- Proteins are categorized as acidic (anions, with acidic amino acids) and basic (cations, abundant in basic amino acids like lysine and arginine).
- What are the vital functions of proteins in the body?
- Proteins serve as enzymes, structural elements (e.g., keratin, collagen), molecular instruments for genetic expression, oxygen and CO2 transport, hemostatic control, defense against infections, and more.
- Which proteins are involved in blood clotting, and how do they function?
- Thrombin, fibrinogen, and other protein factors are involved in blood clotting, forming clots and preventing excessive bleeding.
- What role do proteins play in hereditary transmission, and which proteins are storage proteins?
- Proteins, like nucleoproteins in the cell nucleus, are involved in hereditary transmission. Storage proteins include ovalbumin and glutelin.
- How do actin and myosin function as proteins in the body?
- Actin and myosin serve as contractile proteins crucial for muscle contraction.
- What percentage of the total dry weight of cells do proteins comprise?
- Proteins comprise over 50% of the total dry weight of cells.
Summarizing the Proteins Tutorial
In this comprehensive tutorial, we delved into the diverse aspects of proteins, covering topics such as Peptide Linkage, Structure, Classification, and Functions. Proteins, constituting over 50% of the total dry weight in cells, are crucial macromolecules present in all cell types and parts. Comprising polymers of amino acids, these organic compounds play a pivotal role in various biological processes.
Peptide Linkage
The Peptide Linkage section elucidated how amino acids form polypeptides through the creation of peptide bonds, leading to the development of diverse polypeptide chains.
Structure of Proteins
The Structure of Proteins segment explored the primary, secondary, tertiary, and quaternary structures, elucidating the intricate arrangements of amino acids involving covalent linkages, hydrogen bonds, and more.
Classification of Proteins
Moving on to the Classification of Proteins, the tutorial outlined classifications based on constitution, shape, and nature of molecules. Simple proteins, conjugated proteins, and derived proteins were explained, offering insights into their composition and characteristics.
- On the Basis of Constitution
- Simple proteins: Composed of amino acid residue, yielding constituent amino acids on hydrolysis.
- Conjugated proteins: Integrated with non-protein parts like Nucleoprotein, Phosphoprotein, Lipoprotein, Metalloprotein.
- Derived proteins: Derivatives or degraded products, including Primary and Secondary derived proteins.
- On the Basis of Shape
- Fibrous proteins: Molecules with fibril-like polypeptide chains, insoluble, and performing structural roles.
- Globular proteins: Spherical or ellipsoidal with multiple folded polypeptide chains, soluble, and involved in various functions.
- On the Basis of Nature of Molecules
- Acidic proteins: Exist as anions, consist of acidic amino acids, e.g., blood groups.
- Basic proteins: Exist as cations, abundant in basic amino acids, e.g., lysine, arginine.
Functions of Proteins
The Functions of Proteins section highlighted their vital roles in development, growth, and diverse biological functions, serving as enzymes, structural elements, molecular instruments for genetic expression, and more. Examples of proteins involved in blood clotting, defense against infections, and hereditary transmission were explored.
In essence, this tutorial serves as a comprehensive guide to understanding the fundamental aspects of proteins, providing valuable insights into their composition, structure, classification, and multifaceted functions in the intricate machinery of living organisms.

